- HOME
- Calcium carbonate Museum
- CaCO3 Properties
Water-soluble mineral
As mentioned earlier, because calcium carbonate is a water-soluble mineral, create a singular topography just like Karst. Chemical Properties which CaCO3 dissolve into water is different from other minerals. CaCO3 does not dissolve into tap water, but it is easy to dissolve into acid water. For example, if you blow a breath(it contains CO2) onto calcium solution, you can see white calcium carbonate precipitation. But if you keep blowing the solution become aerated water, then precipitated CO3 dissolve into water again. This is the lime stone encroaching mechanism in nature.
Shape of CaCO3
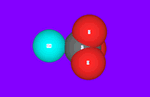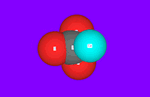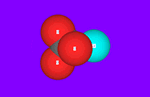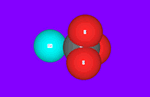
Many calcium carbonate molecules are gathered together, our eyes can catch their shape. A way of gathering determine their properties. This way of gathering is called “Crystal structure”. Calcium carbonate have three types of crystal structures as indicated next table. “Calcite” is most stable crystal structure, so almost lime stone is calcite which out put from mines.
| Calcite | Aragonite | Vaterite | ||
|---|---|---|---|---|
| Crystal structure | Trigonal | Orthorhombic | Hexagonal crystal | |
| Mineral Properties | Hardness | 3 | 3.5 - 4 | 3 |
| A refractive index | 1.49 - 1.69 | 1.53 - 1.69 | 1.56 - 1.66 | |
| Specific gravity | 2.7 | 2.9 | 2.5 - 2.6 |


Source: Celestite Blue HP
(http://www.geocities.jp/celestine_blue_sky/)
(http://www.geocities.jp/celestine_blue_sky/)
High temperature and pressure generated by diastrophism melted a part of coral reef Calcium carbonate. Melted calcium carbonate was cool down very slowly and finally several types of crystals had been formed. Cool down speed and impurities affect the shape of crystals.
CaCO3 groups
| Heavy calcium carbonate | Calcium carbonate which limestone crushed. CaCO3. |
|---|---|
| Precipitated calcium carbonate | A synthetic calcium carbonate reacts with slaked lime and carbon dioxide. CaCO3. |
| Quick lime | Roast limestone and remove CO2 at high temperature(more than 900℃) .CaO |
| Slaked lime | Calcium hydroxide which react with quick lime and water. Ca(OH)2 |